 Through Abbott’s proprietary i2i™ communication technology, AVEIR DR devices provide synchronized or coordinated cardiac pacing between two leadless pacemakers based on the person’s clinical needs. 
On JAbbott announced that the FDA had approved the AVEIR™ dual chamber (DR) leadless pacemaker system, the world’s first dual chamber leadless pacing system. The system, which can be controlled on an Abbott supplied patient controller or a compatible and secure iOS device, offers users helpful notifications and customizable settings for a personalized charging experience”įDA Approves Abbott’s Aveir Dual-Chamber Leadless Pacing System (July 2023) 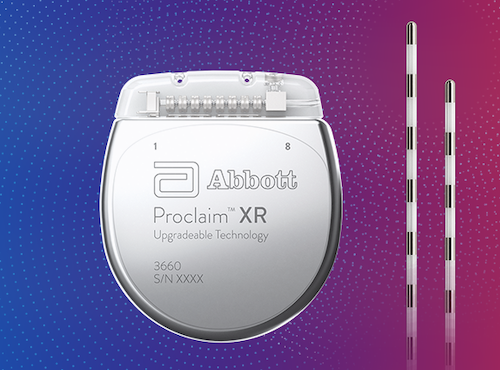
The wireless charging system allows people to be active while wearing it and can fully recharge the Liberta RC DBS system twice before needing to be plugged in again. For users who prefer a weekly charging schedule, only 30 minutes of charging is needed. When used under standard settings, Abbott’s Liberta RC DBS system needs to be recharged as few as every 37 days – or 10 times a year – using a wireless charger that is placed over the device. “At approximately the height and width of a smartwatch face, the Liberta RC DBS system is about 31% smaller than other commonly used implantable, rechargeable DBS devices currently available in the U.S. Jude’s implant was not up to industry standards and that doctors were not warned about the risks of the implant.Abbott announced that it received FDA’s approval for its Liberta RC DBS system, which is the world’s smallest rechargeable deep brain stimulation (DBS) device with remote programming, to treat people living with movement disorders. The Freeds will have an opportunity to amend their warn claim one last time and prove that St. Jude argued that the case be dismissed without prejudice. Burke states that the changes that Freed made to her complaint were enough to support her complaint that the stimulator was not up to industry standards.īurke did dismiss three breach of warranty counts from the lawsuit. The process provides protection to manufacturers so that they don’t have to face state medical device lawsuits.įreed claims that the stimulator was not manufactured up to regulatory and industry standards. Spinal cord stimulators, and similar medical products, are forced to undergo a lengthy, federal approval process. The neurostimulator and its battery components were implanted in the woman’s left buttocks. Jude had to recall similar products in the past. 
Culley, Freed’s attorney, claims that St. Jude responsible for Freed’s pain and suffering.ĭavid G. The judge is allowing the complaint to be revised in an effort to hold St. Jude didn’t warn her or her doctors of the potential risks that are associated with the stimulator. The implant, intended to be permanent, had been implanted at Newark’s Christiana Hospital.įreed has been granted one more opportunity to show that St. She claimed that the stimulator would cause her to suffer from burning and electrical shocks, both of which she claims were very painful. Jude failed to guarantee safety for its spinal cord stimulator.įreed had the stimulator implanted in 2014 and removed less than a year later. The judge ruled against the company after claims that the manufacturer’s spinal cord stimulator was negligently made. 
must defend part of a legal lawsuit against the company, according to a Delaware federal judge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
